“Widespread vaccination against COVID-19 with highly effective vaccines represents an important tool in efforts to control the pandemic,” they wrote. Nevertheless, they said that anaphylaxis appears to be rare. The report’s authors stated that because the available data are limited, it would be difficult to compare the risks of anaphylaxis associated with the Pfizer-BioNTech vaccine with those of non-COVID-19 vaccines. Perhaps most important, no deaths from anaphylaxis were reported after receiving the vaccine. At the time of reporting to VAERS, 20 people had recovered or were discharged home after treatment. 
The investigators also found that the majority of patients who experienced anaphylaxis after receiving the vaccine (81%) had a documented history of allergies or allergic reactions to drugs or other substances, and that one-third had experienced an episode of anaphylaxis in the past.Īs to the seriousness of anaphylaxis, four people required hospitalization and the other 17 were treated in an emergency department. Most people who experienced anaphylaxis (86%) had onset within 30 minutes of receiving the vaccine, and 19 patients were treated with epinephrine as part of therapy. The most common signs and symptoms were a rash and/or hives. Of the 21 individuals in whom anaphylaxis occurred, 19 were women the median age was 40. The overall adverse event rate was slightly more than 0.02%, and the estimated anaphylaxis rate was about 11 cases per million doses administered. seven were still under investigation at the time the report was published.61 were non-allergic adverse events and.86 were non-anaphylaxis allergic reactions,.Of those, 175 were initially identified as possible cases of severe allergic reaction. Methods and AnalysisĪuthors examined 4,393 adverse event reports following administration of the Pfizer-BioNTech vaccine that were submitted to VAERS between Dec. “There was nothing in this report that was particularly concerning but (it) was a good reminder that family physicians must be prepared to recognize early signs of anaphylaxis and be prepared to treat,” Coles said. Sarah Coles, M.D., chair of the AAFP’s Commission on Health of the Public and Science, told AAFP News in an email that for family physicians, the take-home message from the report is that the vaccine is safe and effective, and that anaphylaxis is a rare complication. The report serves to remind family physicians and other health care professionals of the importance of recognizing the signs and symptoms of anaphylaxis in patients who are vaccinated against COVID-19, and also to ensure that clinicians have sufficient amounts of the supplies necessary to manage allergic reactions to vaccination in the clinic or office setting. I have followed all appropriate research reporting guidelines and uploaded the relevant EQUATOR Network research reporting checklist(s) and other pertinent material as supplementary files, if applicable.Those are the main findings from an analysis of VAERS case reports published as a Morbidity and Mortality Weekly Report Early Release on Jan. I confirm that any such study reported in the manuscript has been registered and the trial registration ID is provided (note: if posting a prospective study registered retrospectively, please provide a statement in the trial ID field explaining why the study was not registered in advance). I understand that all clinical trials and any other prospective interventional studies must be registered with an ICMJE-approved registry, such as. The IEC of AIIMS Jodhpur was approached for ethical approval for this manuscript and they have given us exemption from the ethics review.Īll necessary patient/participant consent has been obtained and the appropriate institutional forms have been archived. The details of the IRB/oversight body that provided approval or exemption for the research described are given below: I confirm all relevant ethical guidelines have been followed, and any necessary IRB and/or ethics committee approvals have been obtained. No funding received from any source Author Declarations The authors have declared no competing interest. To link these SAEs to vaccines will need causality analysis and review of individual reports. 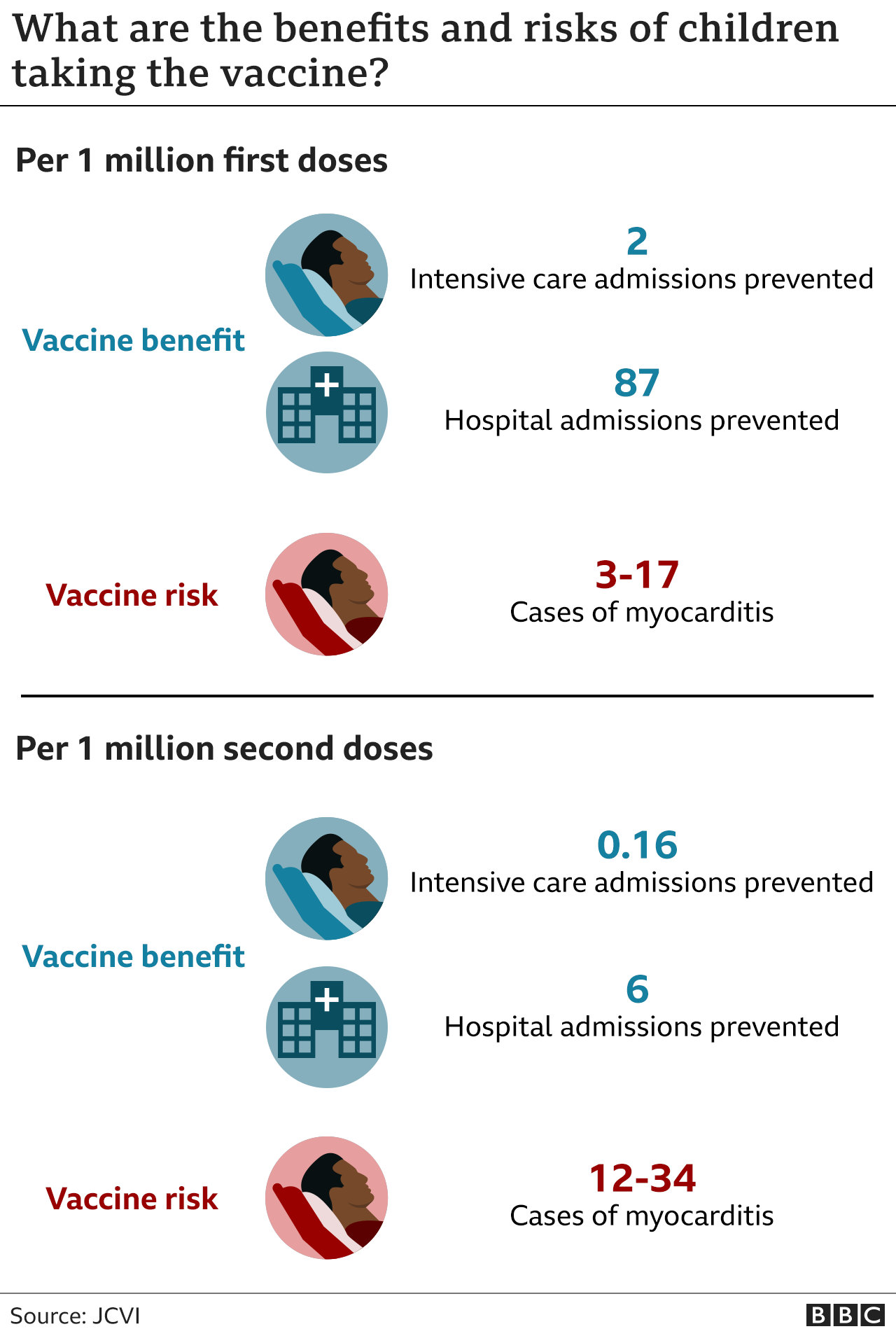
Conclusion The reported SAEs from the COVID–19 vaccines were in line with the data published in clinical trials. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
